LacertaMatrix™:A Flexible Wound Matrix for Clinical Use
LacertaMatrix™:
A Flexible Wound Matrix for Clinical Use
LacertaMatrix™ is a single use, non-pyrogenic, flexible, and conformable wound matrix made from alligator-derived hyaluronic acid (HA) and porcine gelatin.1
The Science
Benefits of American Alligator Hyaluronic Acid (HA)
American Alligator Hyaluronic Acid (HA) is considered beneficial, especially for wound healing, because it can enhance the body’s natural repair process by supporting cell infiltration, hydration, and in promoting tissue regeneration by reducing inflammation in chronic wounds2,3,4,5
Tissue Repair & Regeneration
Studies have shown that HA is crucial for wound healing, supporting cell infiltration (like skin cells), collagen deposition, and blood vessel formation (angiogenesis)2,3,4,5
Cellular Communication
HA has shown to interact with cell surface receptors (like CD44) to influence cell proliferation, differentiation, and immune responses, controlling inflammation.6,7
Modulates Inflammation
HA can form protective “cables” that bind inflammatory cells, preventing excessive tissue damage during immune responses.6,7
Size Matters
Different molecular sizes of HA (high vs. low weight) have distinct roles, with large HA supporting structure and smaller fragments potentially triggering repair, a complex balance.8,9
- LacertaMatrix Instructions For Use (IFU)
- Garantziotis S, Savani RC. Hyaluronan biology: A complex balancing act of structure, function, location and context. Matrix Biol. 2019 May;78-79:1-10. doi: 10.1016/j.matbio.2019.02.002. Epub 2019 Feb 23. PMID: 30802498; PMCID: PMC6774756.
- Matthew Dovedytis, Zhuo Jie Liu, Samuel Bartlett, Hyaluronic acid and its biomedical applications: A review, Engineered Regeneration, Volume 1, 2020, Pages 102-113, ISSN 2666-1381, https://doi.org/10.1016/j.engreg.2020.10.001.
- Mitch Sanders, Mia Hanna, Vanessa Vu, Lindsay Poland. Alligator-Derived Hyaluronic Acid: Bacteriostatic and Fungastatic Properties Against Pathogens. Wound Masterclass Volume 1: Issue 3, December 2022, Pages 60-67.
- Kawano Y, Patrulea V, Sublet E, Borchard G, Iyoda T, Kageyama R, Morita A, Seino S, Yoshida H, Jordan O, Hanawa T. Wound Healing Promotion by Hyaluronic Acid: Effect of Molecular Weight on Gene Expression and In Vivo Wound Closure. Pharmaceuticals (Basel). 2021 Mar 28;14(4):301. doi: 10.3390/ph14040301. PMID: 33800588; PMCID: PMC8065935.
- Toole BP. Hyaluronan-CD44 Interactions in Cancer: Paradoxes and Possibilities. Clin Cancer Res. 2009 Dec 15;15(24):7462-7468. doi: 10.1158/1078-0432.CCR-09-0479. PMID: 20008845; PMCID: PMC2796593.
- Petrey AC, de la Motte CA. Hyaluronan, a crucial regulator of inflammation. Front Immunol. 2014 Mar 11;5:101. doi: 10.3389/fimmu.2014.00101. PMID: 24653726; PMCID: PMC3949149.
- Tavianatou AG, Caon I, Franchi M, Piperigkou Z, Galesso D, Karamanos NK. Hyaluronan: molecular size-dependent signaling and biological functions in inflammation and cancer. FEBS J. 2019 Aug;286(15):2883-2908. doi: 10.1111/febs.14777. Epub 2019 Feb 21. PMID: 30724463.
- Ruppert SM, Hawn TR, Arrigoni A, Wight TN, Bollyky PL. Tissue integrity signals communicated by high-molecular weight hyaluronan and the resolution of inflammation. Immunol Res. 2014 May;58(2-3):186-92. doi: 10.1007/s12026-014-8495-2. PMID: 24614953; PMCID: PMC4106675.
Tissue Repair & Regeneration
Studies have shown that HA is crucial for wound healing, supporting cell infiltration (like skin cells), collagen deposition, and blood vessel formation (angiogenesis)2,3,4,5
Cellular Communication
HA has shown to interact with cell surface receptors (like CD44) to influence cell proliferation, differentiation, and immune responses, controlling inflammation.6,7
Modulates Inflammation
HA can form protective “cables” that bind inflammatory cells, preventing excessive tissue damage during immune responses.6,7
Size Matters
Different molecular sizes of HA (high vs. low weight) have distinct roles, with large HA supporting structure and smaller fragments potentially triggering repair, a complex balance.8,9
- LacertaMatrix Instructions For Use (IFU)
- Garantziotis S, Savani RC. Hyaluronan biology: A complex balancing act of structure, function, location and context. Matrix Biol. 2019 May;78-79:1-10. doi: 10.1016/j.matbio.2019.02.002. Epub 2019 Feb 23. PMID: 30802498; PMCID: PMC6774756.
- Matthew Dovedytis, Zhuo Jie Liu, Samuel Bartlett, Hyaluronic acid and its biomedical applications: A review, Engineered Regeneration, Volume 1, 2020, Pages 102-113, ISSN 2666-1381, https://doi.org/10.1016/j.engreg.2020.10.001.
- Mitch Sanders, Mia Hanna, Vanessa Vu, Lindsay Poland. Alligator-Derived Hyaluronic Acid: Bacteriostatic and Fungastatic Properties Against Pathogens. Wound Masterclass Volume 1: Issue 3, December 2022, Pages 60-67.
- Kawano Y, Patrulea V, Sublet E, Borchard G, Iyoda T, Kageyama R, Morita A, Seino S, Yoshida H, Jordan O, Hanawa T. Wound Healing Promotion by Hyaluronic Acid: Effect of Molecular Weight on Gene Expression and In Vivo Wound Closure. Pharmaceuticals (Basel). 2021 Mar 28;14(4):301. doi: 10.3390/ph14040301. PMID: 33800588; PMCID: PMC8065935.
- Toole BP. Hyaluronan-CD44 Interactions in Cancer: Paradoxes and Possibilities. Clin Cancer Res. 2009 Dec 15;15(24):7462-7468. doi: 10.1158/1078-0432.CCR-09-0479. PMID: 20008845; PMCID: PMC2796593.
- Petrey AC, de la Motte CA. Hyaluronan, a crucial regulator of inflammation. Front Immunol. 2014 Mar 11;5:101. doi: 10.3389/fimmu.2014.00101. PMID: 24653726; PMCID: PMC3949149.
- Tavianatou AG, Caon I, Franchi M, Piperigkou Z, Galesso D, Karamanos NK. Hyaluronan: molecular size-dependent signaling and biological functions in inflammation and cancer. FEBS J. 2019 Aug;286(15):2883-2908. doi: 10.1111/febs.14777. Epub 2019 Feb 21. PMID: 30724463.
- Ruppert SM, Hawn TR, Arrigoni A, Wight TN, Bollyky PL. Tissue integrity signals communicated by high-molecular weight hyaluronan and the resolution of inflammation. Immunol Res. 2014 May;58(2-3):186-92. doi: 10.1007/s12026-014-8495-2. PMID: 24614953; PMCID: PMC4106675.
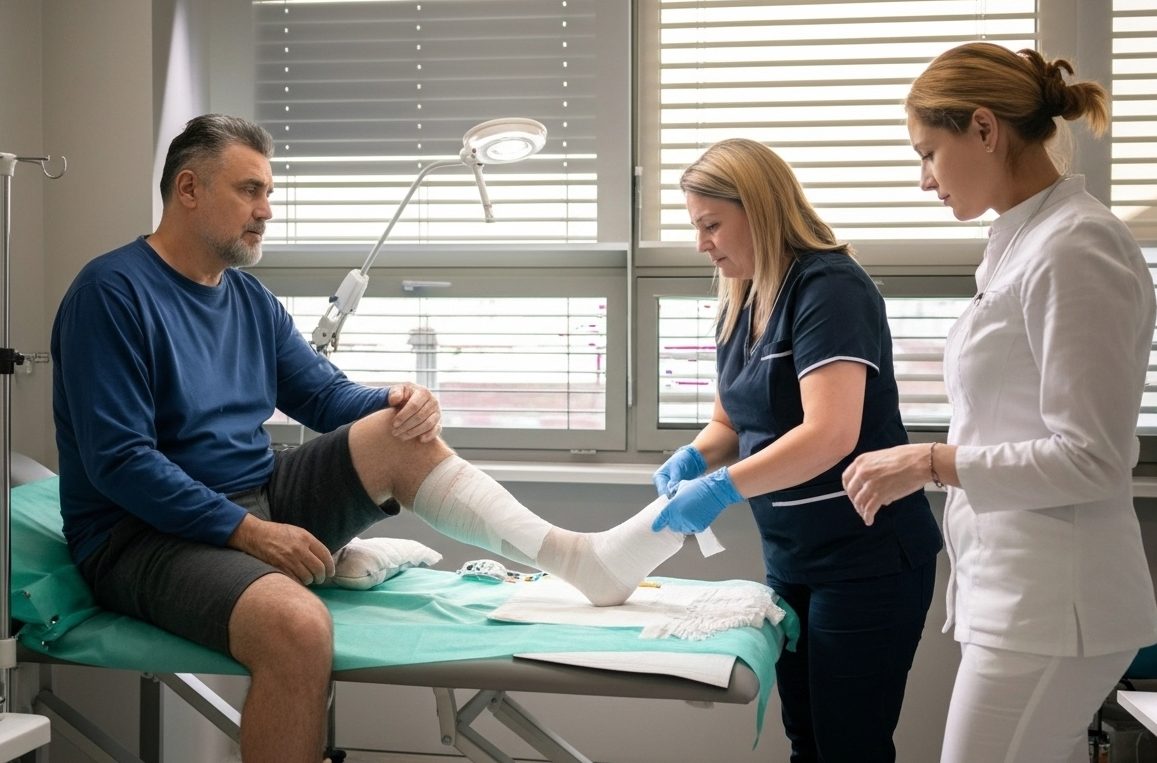
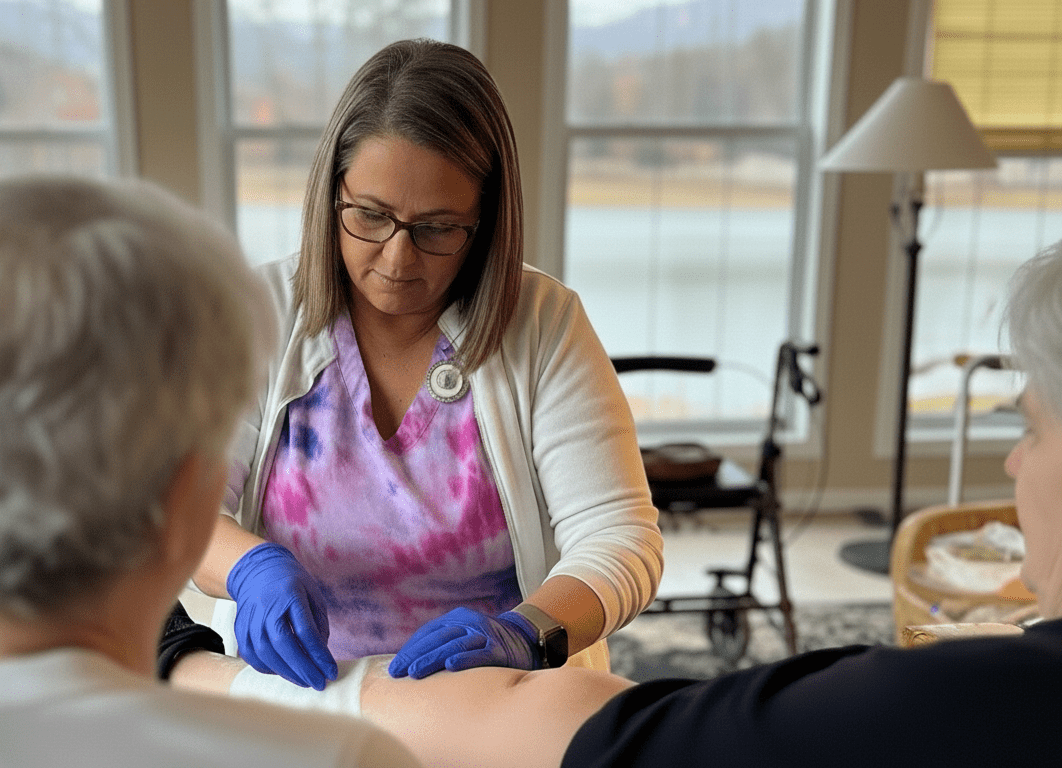
Clinical Use
Intended Wound Types
LacertaMatrix™ is intended for use across a broad range of wound types.
This includes surgical wounds, such as donor sites, grafts, post-Moh’s surgery wounds, post-laser surgery wounds, podiatric wounds, and wound dehiscence.
LacertaMatrix™ may also be used for trauma wounds, including abrasions, lacerations, partial thickness burns, and skin tears, as well as draining wounds.
Commonly Managed Wound Types
- Pressure ulcers
- Venous ulcers
- Partial and full thickness wounds
- Diabetic ulcers
- Chronic vascular ulcers
- Tunneled or undermined wounds
- Pressure ulcers
- Venous ulcers
- Partial and full thickness wounds
- Diabetic ulcers
- Chronic vascular ulcers
- Tunneled or undermined wounds
Application
LacertaMatrix™: Application Highlights
Size Selection
Select a device size slightly larger than the wound. LacertaMatrix™ may be applied as a whole sheet or trimmed to contact wound margins.
Preparation & Handling
Inspect the packaging to ensure it is intact and undamaged. Using aseptic technique, remove the device from the double-pouch configuration.
Placement & Conformity
For best results, ensure LacertaMatrix™ conforms to the underlying wound bed and is placed in maximum contact with healthy, well-vascularized tissue.
Resorption Over Time
LacertaMatrix™ resorbs into the wound over time, typically over a period of approximately two weeks.
Secondary Dressing
Protect LacertaMatrix™ using an appropriate secondary dressing based on wound characteristics and clinician preference.
Ongoing Care
Change the secondary dressing as needed to maintain a moist, clean wound area and allow wound inspection.
Size Selection
Preparation & Handling
Placement & Conformity
Resorption Over Time
Secondary Dressing
Ongoing Care
Note: Do not use the device in patients with a known sensitivity to materials of porcine (pig) or reptilian (alligator) origin.
Specifications
Product Sizes
| Part Number / SKU | Device Size | Size (SQ CMs) |
|---|---|---|
| LAC-011 | LacertaMatrix™ 1cm x 1cm | 1 |
| LAC-012 | LacertaMatrix™ 1cm x 2cm | 2 |
| LAC-013 | LacertaMatrix™ 1cm x 3cm | 3 |
| LAC-025 | LacertaMatrix™ 2.5cm x 2.5cm | 7 |
| LAC-024 | LacertaMatrix™ 2cm x 4cm | 8 |
| LAC-033 | LacertaMatrix™ 3cm x 3cm | 9 |
| LAC-034 | LacertaMatrix™ 3cm x 4cm | 12 |
| LAC-044 | LacertaMatrix™ 4cm x 4cm | 16 |
| LAC-050 | LacertaMatrix™ 5cm x 5cm | 25 |
| LAC-048 | LacertaMatrix™ 4cm x 8cm | 32 |
| LAC-088 | LacertaMatrix™ 8cm x 8cm | 64 |
| LAC-100 | LacertaMatrix™ 10cm x 10cm | 100 |